Introduction: In the phase 3 ECHELON-2 study (NCT01777152), treatment with brentuximab vedotin (BV) + cyclophosphamide, doxorubicin, and prednisone (A+CHP) demonstrated significantly longer progression-free and overall survival compared with cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) in the frontline (FL) treatment of patients with systemic anaplastic large cell lymphoma (sALCL) or other CD30-expressing peripheral T-cell lymphomas (PTCL). This study supported the November 2018 US FDA approval of A+CHP as FL therapy for adults with sALCL or other CD30-expressing PTCL. The current analysis describes patient characteristics, PTCL subtypes, and supportive care use of FL A+CHP and CHOP outside of the clinical trial setting in the US.
Methods: Using medical and pharmacy claims data in the Symphony Health Solutions database, a retrospective cohort analysis of patients with PTCL treated with FL A+CHP or CHOP was conducted to compare treatment and utilization characteristics. Patients ≥18 years with 1 inpatient or 2 outpatient ICD-9/10 PTCL diagnosis codes, newly initiated on A+CHP or CHOP (index date) between November 2018 and January 2020, and with ≥6 months continuous activity before and ≥3 months after the index date were included. To adjust for confounding factors, a 1:1 propensity score matching analysis was performed based on age, gender, baseline comorbidities, geographic region and length of follow-up.
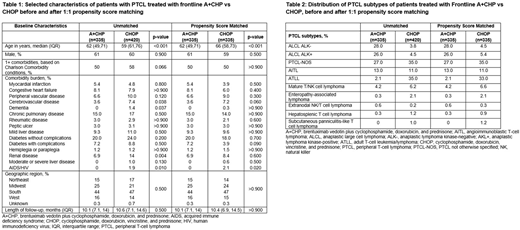
Results: A total of 755 patients met inclusion criteria (335 A+CHP; 420 CHOP) with a median follow-up period of 10.1 and 10.6 months, respectively. In the unmatched cohorts, 61% were male, and median age at index was 62 and 69 years for A+CHP and CHOP, respectively. The prevalence of comorbidities based on the Charlson Comorbidity Index was similar between the cohorts; prevalent conditions included diabetes, chronic pulmonary disease, congestive heart failure, and liver disease (Table 1). PTCL subtypes treated with A+CHP included sALCL (54%), PTCL-not otherwise specified (NOS; 27%), and angioimmunoblastic T-cell lymphoma (AITL; 13%); subtypes treated with CHOP included PTCL-NOS (35%), adult T-cell leukemia/lymphoma (ATLL; 35%), and AITL (11%) (Table 2). After matching, the proportion of patients who received granulocyte-colony stimulating factor (G-CSF; A+CHP: 91%, CHOP: 86%, p=0.1) and the incidence rate of neutropenia (A+CHP: 45%, CHOP: 42%, p=0.4) during FL treatment for both study cohorts was similar. Of patients who received G-CSF, the majority received it as primary prophylaxis given within the first 5 days of FL treatment initiation (A+CHP: 89%, CHOP: 85%, p=0.2). The rate of subsequent therapy (ie, therapy change after FL), was similar between A+CHP and CHOP (18% vs 21%; p=0.3) and for the sALCL subtype (16% vs 26%, p=0.2). Of the A+CHP patients who received subsequent therapy, 32% were retreated with a BV-containing regimen and 19% of CHOP patients received a BV-containing regimen.
Conclusions: In this real-world analysis, US patients with PTCL newly initiated on A+CHP or CHOP were older (67 vs 58 years) than those in ECHELON-2. There was a high comorbidity burden; over half of the patients in both cohorts had 1+ comorbidities, a potential reflection of the older population. As would be expected due to a high rate of CD30-positivity in the disease, A+CHP was more commonly used than CHOP in sALCL. In PTCL subtypes in which CD30 is more variably expressed, A+CHP and CHOP were used with similar frequencies. Although clinical trials in ATLL have demonstrated improved outcomes with more complex and intensive regimens than CHOP, CHOP remains commonly used in ATLL. A+CHP was also used in PTCL subtypes not included in ECHELON-2, such as NK/T cell lymphomas. G-CSF was used as primary prophylaxis in the large majority of patients in both cohorts. The use of a BV-containing regimen as subsequent therapy was more common in A+CHP vs CHOP, probably because the tumors of A+CHP patients were more likely to have expressed CD30. Confounding by unmeasured characteristics cannot be ruled out due to inherent limitations in claims data (eg, lack of disease stage, CD30 testing and response outcomes). Characteristics and management of this real-world population with PTCL differed from those in the ECHELON-2 trial, demonstrating the importance of retrospective studies to assess the impact of new regimens on clinical practice and to identify areas for further education of practitioners.
Burke:Seattle Genetics: Speakers Bureau; Gilead: Consultancy; Bristol Myers Squibb: Consultancy; Roche: Consultancy; Epizyme: Consultancy; Adaptive: Consultancy; Kura: Consultancy; Morphosys: Consultancy; Celgene: Consultancy; Adaptive Biotechnologies: Consultancy; Verastem: Consultancy; Astra Zeneca: Consultancy; Bayer: Consultancy; AbbVie: Consultancy. Liu:Seattle Genetics: Current Employment, Current equity holder in publicly-traded company. Yu-Isenberg:Seattle Genetics: Current Employment, Current equity holder in publicly-traded company. Fanale:Seattle Genetics: Current Employment, Current equity holder in publicly-traded company. Surinach:Seattle Genetics: Research Funding. Flores:Seattle Genetics: Research Funding. Lisano:Seattle Genetics: Current Employment, Current equity holder in publicly-traded company. Phillips:Beigene: Consultancy; AstraZeneca: Consultancy; Karyopharm: Consultancy; Bayer: Consultancy, Research Funding; Lymphoma Connect: Other; Incyte: Consultancy, Research Funding; Cardinal Health: Consultancy; University of Michigan: Current Employment; Pharmacyclics: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding; BMS: Consultancy; Seattle Genetics: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal